|
From your observations label them as metal, non-metal or metalloid. On the blank periodic table, label the elements that we tested in lab. (Note: not all reactions are vigorous, so watch closely)Įlement Appearance Conductivity Malleability Reactivity with HCL Non-metal Metal or Metalloidįor the following elements, try to decide if they are a metal, non-metal or metalloid based on their appearance.Įlement Appearance Non-metal Metal or Metalloid Formation of bubbles of hydrogen is evidence that a reaction is occurring. Then add a small sample (approx 0.1 gram) of each element to the labeled tubes. To test the reactivity with 1 M HCl, label 9 test tubes with the symbols for each element. An element is malleable if it flattens when it is tapped.ĥ. An element is brittle if it shatters when it is hit. To determine which elements are malleable, place a single piece of the element on a paper towel, and gently tap it with a hammer. If you observe carefully, you might see that some are semi-conductors.Ĥ. Using the micro-conductivity tester, determine whether the elements conduct electricity. Record physical state, color, luster and other observable characteristics.ģ. Observe the appearance of each of the elements. In your lab notebook, draw a table like the one shown below.Ģ. Write the whole property, not just the letter of the property.įor the observation stations you should prepare sealed test tubes containing the following elements:Ĭopper, silicon, magnesium, carbon, nickel, aluminum, zinc, sulfur, oxygen, lead, bismuth, silver, nitrogen, antimony, and hydrogenįor the conductivity station, you should have plastic dishes with one piece of each element to be tested and micro-conductivity testers.įor the malleability station, have one piece of each element per group along with paper towels and hammers.įor the reactivity station, have one piece of each element per group, 9 test tubes, test tube rack and 1 M HCl.ġ. Write each of the following properties under the appropriate heading. In you lab notebook, draw the following table. To investigate the properties of several elements on the periodic table and classify them as metals, non-metals or metalloids. A copy is also attached in the documents. This is a copy of the student handout of the lab. 
After everyone has completed the write-up, we will compare our results with the book classification and location of these elements. 
They will use this information to answer the analysis questions at the end of the lab. 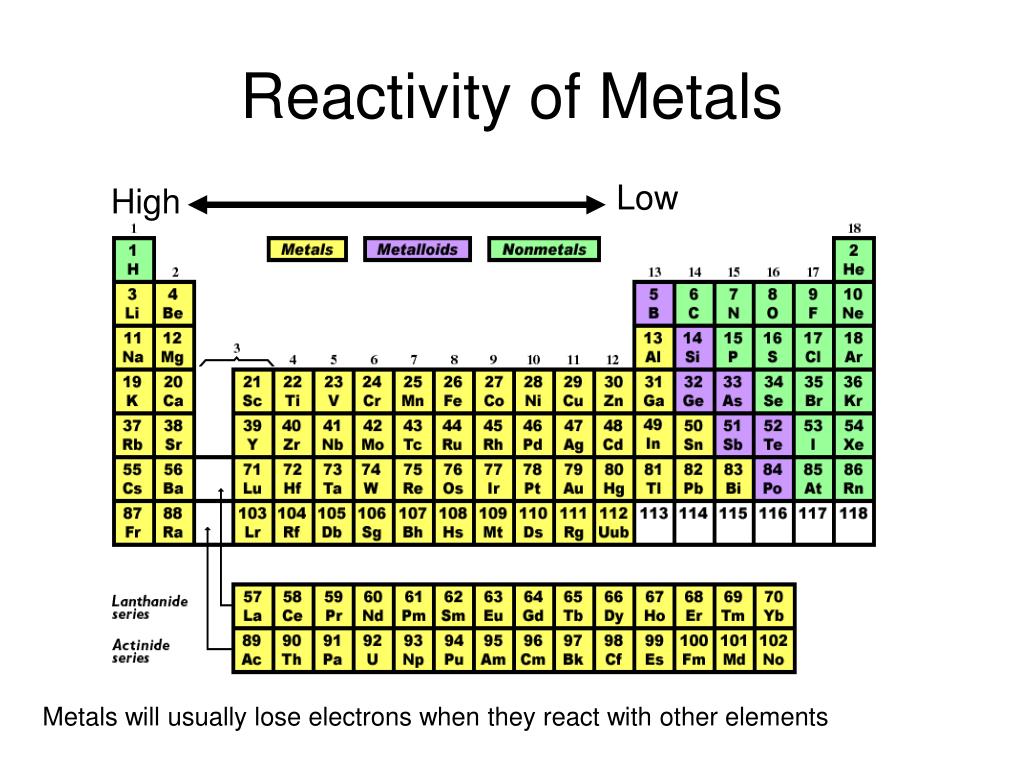 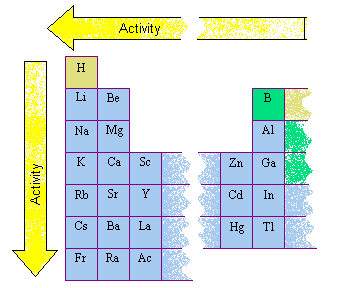
They will label each of these elements on a blank periodic table and color each group a different color. After they have completed their observations, they will determine if each element that they tested is a metal, non-metal or metalloid based on their observations. The stations can be completed in any order. Students will work in pairs or small groups and will rotate through the observation, conductivity, malleability and reactivity stations until they have completed all the stations. Students will be given the student handout (attached) and asked to write the experiment up in their lab notebooks. Before starting the lab, we will review the properties of each group. This lab activity will be used to have students discover the location of metals, non-metals and metalloids on the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
